Proper nutrition can accelerate injury recovery and help maintain lean mass during times of muscle disuse. In this article, I review nutritional strategies to get you back into action more quickly.
Injury Stages
Recovery from musculoskeletal injury occurs in two phases. Phase 1, healing and recovery, is characterized by the wound-healing process after injury. This involves an inflammatory response followed by tissue remodeling. Phase 2, returning to activity, involves increased usage and rehabilitation of the immobilized limb(s). We will focus on nutritional strategies during Phase 1, the healing and recovery from a musculoskeletal injury.
Goals for Recovery from Injury
- Prevent chronic, excessive inflammation.
- Prevent muscle loss.
Inflammation is a natural response induced by a musculoskeletal injury. Although inflammation is counterproductive to recovery, this natural inflammatory response is necessary and integral for proper wound healing to occur [1-3].
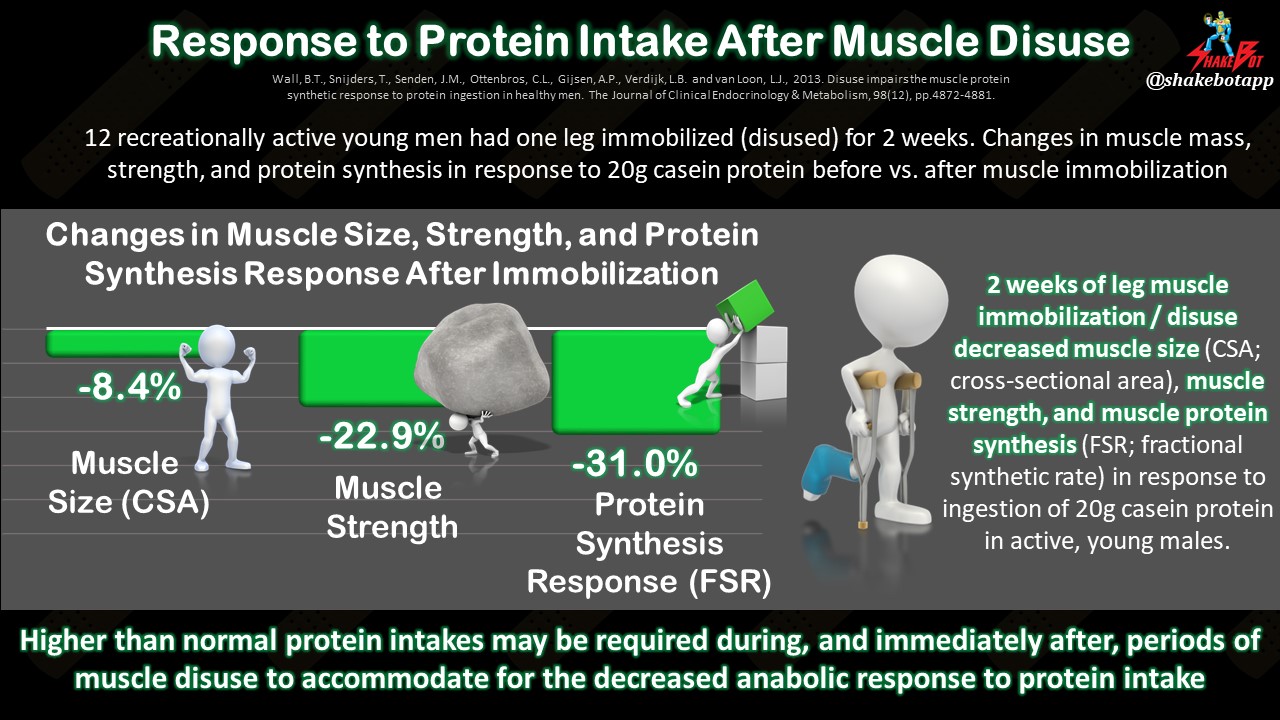
Typically, an injury will result in muscle disuse. When a muscle isn’t being used, it quickly loses strength and function [4-8]. The body’s ability to build new protein is also compromised during injury. This combination of 1) a decrease in muscle use, and 2) the body’s reduced response to protein ingestion, causes muscle atrophy, or muscle loss [7, 9]. When muscles are inactive, as is the case during injury, muscle tissue lessens by approximately 0.5% per day [11, 12], with the first two weeks of inactivity stimulating the greatest proportion of relative muscle loss [10]. The two primary nutrition goals for injury recovery are to (1) prevent inflammation after the initial, natural inflammatory response, and (2) mitigate muscle losses.
Primary Consideration 1: Energy Balance
When injured, a substantial decrease in physical activity and training is common, which significantly reduces energy expenditure (calories burned during the day). One may think that cutting the cord on food should balance things out. This would be a huge mistake and would undoubtedly lead to a lengthened injury period. The healing process actually causes an increase in resting energy expenditure by 15-50%, depending on the type and severity of the injury [13]. This increase in caloric expenditure is often overlooked, but needs to be considered when determining energy needs in light of the decreased physical activity (and thus physical caloric expenditure) that coincides with injury. Ensuring that adequate energy requirements are met is crucial during this time; not only will an energy deficit interfere with wound healing [13], but it may also exacerbate muscle losses [14, 15]. On the flip side of the coin, a significant energy surplus may cause systemic inflammation [16] and can result in unwanted gains in body fat [17].
- Energy restriction = ? Wound healing, ? Muscle loss
- Energy surplus = ? Wound healing (paucity of research, currently), ? Fat gain
A primary nutritional goal of injury recovery should be to make appropriate nutritional choices to maintain energy balance, rather than supporting a state of excessive energy intake, or restriction.
Primary Consideration 2: Protein Intake
Insufficient protein intake will increase inflammation, inhibit wound healing, [18, 19] and induce muscle loss via decreases in muscle protein synthesis (MPS) rates [20-22]. To reduce muscle losses and speed up recovery, keep daily protein intake high [19]; between 1.8-2.5g protein per kilogram of bodyweight per day should suffice for most individuals.
In younger individuals, 20-25g of protein in a meal has been shown to maximize MPS [22, 23], whereas older individuals (>65 years) may require more; 35-40g of protein per meal [24, 25]. Since immobilization and/or reduced physical activity causes anabolic resistance (i.e. decreased response to protein intake), additional protein per meal may be required to maximally stimulate MPS during injury periods [25]. To maximize your daily MPS response (i.e. ability to build protein), spread your protein intake into several 20-40 gram servings throughout the day rather than consuming few, high-protein servings [26, 27]. Summarized protein intake recommendations are below:
- 1.8-2.5 g protein/kg of bodyweight/day
- 3-8 meals of 20-40g high-quality protein per meal, dispersed evenly throughout the day
Primary Consideration 3: Omega-3 Fatty Acids
As mentioned previously, one of the primary nutritional goals for injury recovery is to limit inflammation. Since Omega-3 fatty acids are anti-inflammatory, one would assume that shoveling heaps of Omega-3s into the body would speed up injury recovery. Not so fast. There is evidence that supplementation with Omega-3s can actually impair wound healing [28, 29]. However, high Omega-3 intake has also been suggested to reduce muscle losses associated with inactivity [30-32]. So, Omega-3s are detrimental to injury recovery in that they impair wound healing, but are also beneficial in that they will mitigate muscle losses? This situation gets a bit tricky. It would be wise to keep Omega-3 intake low immediately following injury so the natural inflammatory process is undisturbed. After the initial inflammatory response subsides (2-5 days following injury), increasing Omega-3 intake may help support muscle strength and function. It’s important to note that the characteristics of Omega-3 supplementation designed to optimally reduce inflammation, such as the dose, frequency and duration of intake of nutrients, are currently unknown. There is some research supporting the efficacy of Omega-3 supplementation, in certain situations, such as following traumatic brain injury, but the body of evidence is limited. Nutrition and supplementation for brain-related pathology is beyond the scope of this article; the insights included in this article are regarding musculoskeletal injury, only. Given this information, I advise the following guidelines:
- Keep Omega-3 fatty acid intake low for the initial 2-5 days following injury onset to promote proper wound healing
- Increase Omega-3 fatty acid intake to 4-6g per day thereafter to support muscle maintenance once the initial inflammatory process has conceded (hopefully).
Primary Consideration 4: Avoid micronutrient deficiencies and alcohol
There are plenty of micronutrients involved with the healing process as well as MPS, including zinc, vitamin C, vitamin A, and vitamin D, to name a few [33, 34]. However, research is inconclusive; there isn’t a substantial body of evidence that suggests supranormal intake of any of these nutrients will accelerate injury recovery. With that being said, deficiencies of these nutrients can leave the door open for suboptimal recovery [18]. Therefore, we advise that you consume satisfactory amounts of these nutrients, but don’t overdo it. Consuming a varied, colorful diet is advised, but you probably shouldn’t add multiple vitamin/mineral supplements on top of that. Additionally, alcohol impairs wound healing and increases muscle loss. Alcohol consumption should be avoided at all costs when recovering from injury [35-38].

Practical Applications
- Maintain energy balance; weigh yourself frequently under identical conditions to ensure you aren’t gaining or losing weight.
- Keep protein intake high; eat 1.8-2.5 g/kg of bodyweight/day, consisting of multiple 20-40g protein meals dispersed throughout the day.
- Do not supplement with fish oil or omega-3 fatty acids until after the first 3-5 days following injury; although research is lacking in the area, it may be appropriate to supplement with 4-6g per day or ingest high quantities of omega-3 fatty acid sources (ex: seafood, flaxseed oil, walnuts, spinach) after the initial post-injury inflammatory process has concluded.
- Eat a varied, colorful diet to avoid nutrient deficiencies.
- Avoid alcohol intake at all costs; it’s detrimental to recovery.
Reference
- Lorenz HP, Longaker MT. Wounds: Biology, Pathology, and Management. In: Norton JA, Barie PS, Bollinger RR, Chang AE, Lowry SF,Mulvhill SJ, et al., editors. Surgery: basic science and clinical evidence. 2nd ed. New York: Spring Publishing Company; 2008. p. 191–208.
- Stechmiller JK. Understanding the role of nutrition and wound healing. Nutr Clin Pract. 2010;25:61–8.
- Lin E, Kotani JG, Lowry SF. Nutritional modulation of immunity and the inflammatory response. Nutrition. 1998;14:545–50.
- Malliaropoulos N, Papacostas E, Kiritsi O, et al. Posterior thigh muscle injuries in elite track and field athletes. Am J Sports Med. 2010;38:1813–9.
- Ferrando AA, Lane HW, Stuart CA, et al. Prolonged bed rest decreases skeletal muscle and whole body protein synthesis. Am J Physiol. 1996;270:E627–33.
- Ferrando AA, Stuart CA, Brunder DG, et al. Magnetic resonance imaging quantitation of changes in muscle volume during 7 days of strict bed rest. Aviat Space Environ Med. 1995;66:976–81.
- Glover EI, Phillips SM, Oates BR, et al. Immobilization induces anabolic resistance in human myofibrillar protein synthesis with low and high dose amino acid infusion. J Physiol. 2008;586:6049–61.
- Wall BT, Dirks ML, Snijders T, et al. Substantial skeletal muscle loss occurs during only 5 days of disuse. Acta Physiol. 2014;210:600 11.
- Gibson JN, Halliday D, Morrison WL, et al. Decrease in human quadriceps muscle protein turnover consequent upon leg immobilization. Clin Sci. 1987;72:503–9.
- Wall, B., Dirks, M., & van Loon, L. (2013). Skeletal muscle atrophy during short-term disuse: Implications for age-related sarcopenia. Ageing Research Reviews, 12, 898–
- Wall, B. T., Dirks, M. L., Verdijk, L. B., Snijders, T., Hansen, D., Vranckx, P., … van Loon, L. J. C. (2012). Neuromuscular electrical stimulation increases muscle protein synthesis in elderly.
- Phillips, S., Glover, E., & Rennie, M. (2009). Alterations of protein turnover underlying disuse atrophy in human skeletal muscle. Journal of Applied Physiology, 107, 645–654. type 2 diabetic men. AJP: Endocrinology and Metabolism, 303, E614–E623.
- Frankenfield D. Energy expenditure and protein requirements after traumatic injury. Nutr Clin Pract. 2006;21:430–7.
- Biolo G, Ciocchi B, Stulle M, et al. Calorie restriction accelerates the catabolism of lean body mass during 2 wk of bed rest. Am J Clin Nutr. 2007;86:366–72.
- Mettler S, Mitchell N, Tipton KD. Increased protein intake reduces lean body mass loss during weight loss in athletes. Med Sci Sports Exerc. 2010;42:326–37.
- Biolo G, Agostini F, Simunic B, et al. Positive energy balance is associated with accelerated muscle atrophy and increased erythrocyte glutathione turnover during 5 wk of bed rest. Am J Clin Nutr. 2008;88:950–8.
- Myer, G.D., Faigenbaum, A.D., Foss, K.B., Xu, Y., Khoury, J., Dolan, L.M., McCambridge, T.M. and Hewett, T.E., 2013. Injury initiates unfavourable weight gain and obesity markers in youth. Br J Sports Med, pp.bjsports-2012.
- Arnold M, Barbul A. Nutrition and wound healing. Plast Reconstr Surg. 2006;117:42S–58S.
- Demling RH. Nutrition, anabolism, and the wound healing process: an overview. Eplasty. 2009;9:e9.
- Glover EI, Phillips SM, Oates BR, et al. Immobilization induces anabolic resistance in human myofibrillar protein synthesis with low and high dose amino acid infusion. J Physiol. 2008;586:6049–61.
- Biolo G, Tipton KD, Klein S, et al. An abundant supply of amino acids enhances the metabolic effect of exercise on muscle protein. Am J Physiol. 1997;273:E122–9.
- Witard OC, Jackman SR, Breen L, et al. Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. Am J Clin Nutr. 2014;99:86–95.
- Moore DR, Robinson MJ, Fry JL, et al. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am J Clin Nutr. 2008;89:161–8.
- Pennings, B., Boirie, Y., Senden, J., Gijsen, A., Kuipers, H., & van Loon, L. (2011). Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. American Journal of Clinical Nutrition, 93, 997–1005.
- Yang, Y., Breen, L., Burd, N. A., Hector, A. J., Churchward-Venne, T. A., Josse, A. R., … Phillips, S. M. (2012). Resistance exercise enhances myofibrillar protein synthesis with graded intakes of whey protein in older men. British Journal of Nutrition, 7, 1–9.
- Areta JL, Burke LM, Ross ML, et al. Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J Physiol. 2013;591:2319–31.
- Mamerow MM, Mettler JA, English KL, et al. Dietary protein distribution positively influences 24-h muscle protein synthesis in healthy adults. J Nutr. 2014;144:876–80.
- Albina, J.E., Gladden, P. and Walsh, W.R., 1993. Detrimental Effects of an ??3 Fatty Acid?Enriched Diet on Wound Healing. Journal of Parenteral and Enteral Nutrition, 17(6), pp.519-521.
- Otranto M, Do Nascimento AP, Monte-Alto-Costa A. Effects of supplementation with different edible oils on cutaneous wound healing. Wound Repair Regen. 2010;18:629–36.
- You J-S, Park M-N, Song W, et al. Dietary fish oil alleviates soleus atrophy during immobilization in association with Akt signaling to p70s6k and E3 ubiquitin ligases in rats. Appl Physiol Nutr Metab. 2010;35:310–8.
- Smith, G. I., Atherton, P., Reeds, D. N., Mohammed, B. S., Rankin, D., Rennie, M. J., & Mittendorfer, B. (2011a). Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: A randomized controlled trial. American Journal of Clinical Nutrition, 93, 402–412.
- Smith, G., Atherton, P., Reeds, D. N., Mohammed, B. S., Rankin, D., Rennie, M., & Mittendorfer, B. (2011b). Omega-3 polyunsaturated fatty acids augment the muscle protein anabolic response to hyperinsulinaemia–hyperaminoacidaemia in healthy young and middle-aged men and women. Clinical Science, 121, 267–278.
- Barker T, Martins TB, Hill HR, et al. Low vitamin D impairs strength recovery after anterior cruciate ligament surgery. J Evid Based Complement Altern Med. 2011;16:201–9.
- Tipton, K.D. Nutritional support for exercise-induced injuries. Sports Med. 2015, 45, S93–S104.
- Lang CH, Frost RA, Kumar V, et al. Impaired protein synthesis induced by acute alcohol intoxication is associated with changes in eIF4E in muscle and eIF2B in liver. Alcohol Clin Exp Res. 2000;24:322–31.
- Parr EB, Camera DM, Areta JL, et al. Alcohol ingestion impairs maximal post-exercise rates of myofibrillar protein synthesis following a single bout of concurrent training. PLoS One. 2014;9:e88384.
- Jung MK, Callaci JJ, Lauing KL, et al. Alcohol exposure and mechanisms of tissue injury and repair. Alcohol Clin Exp Res. 2011;35:392–9.
- Vargas R, Lang CH. Alcohol accelerates loss of muscle and impairs recovery of muscle mass resulting from disuse atrophy. Alcohol Clin Exp Res. 2008;32:128–37.